|
|
Post by Darren on Oct 7, 2017 18:08:16 GMT
In this article posted by Bloomberg, it's said that the number of new drug approvals are set to increase, with a reduced time from submission to approval. So far, at least nine decisions (from 34) came more than 20 days ahead of the FDA’s scheduled action date. For Iclaprim, the action date is already reduced from the usual 9 months to just 6. 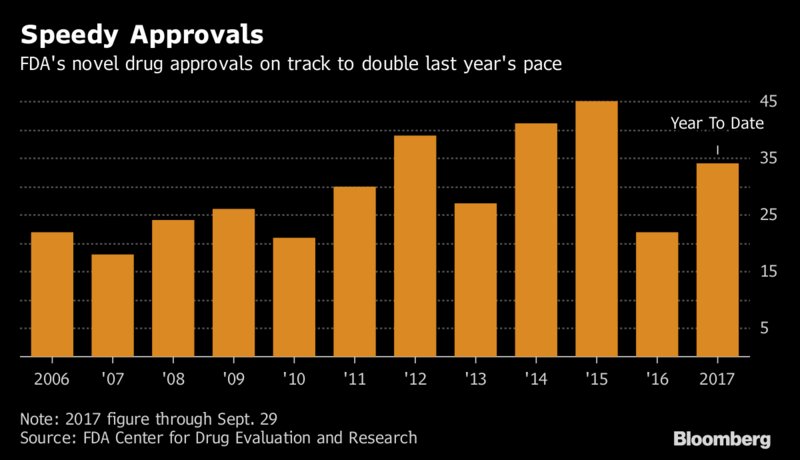 |
|